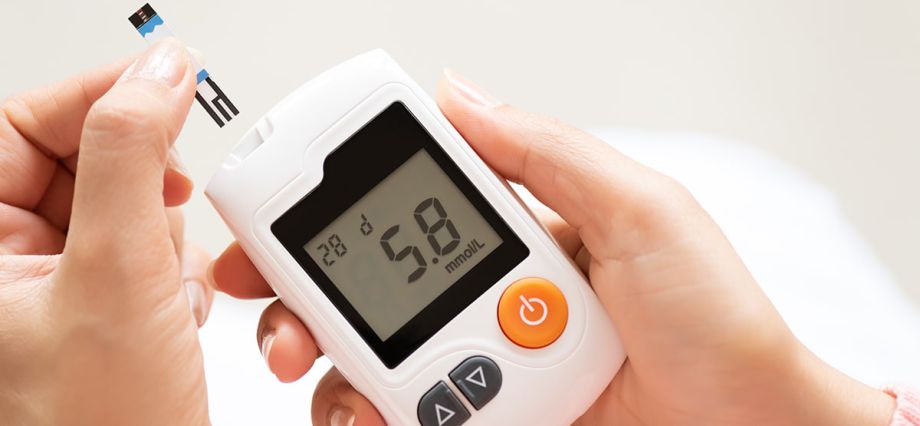
Unique Device Identification System
The FDA established the unique device identification system to adequately identify medical devices sold in the United States from manufacturing through distribution to patient use. When fully implemented,...
FDA requires medical device labelers, GUDID, Medical Devices, Medical Devices Labeling, UDI, Unique Device Identification System